Digital Biomarkers Market Set to Reach US$ 26.69 Billion by 2033, Led by North America's 58.45% Market Share
Digital biomarkers are transforming clinical research and care delivery, enabling real-time, data-driven insights for earlier diagnosis, personalized treatment, and remote patient monitoring.”
AUSTIN, TX, UNITED STATES, February 27, 2026 /EINPresswire.com/ -- The Global Digital Biomarkers Market reached US$ 4.16 Billion in 2024 and is expected to reach US$ 26.69 Billion by 2033, growing at a CAGR of 22.7% during the forecast period 2025-2033.— DataM Intelligence
Market growth is driven by the surging demand for remote patient monitoring, integration of AI and wearables in healthcare, and rising chronic disease prevalence like diabetes and cardiovascular conditions. Advancements in sensor technology, expanding use in clinical trials for real-world evidence, growing investments in digital health platforms, and supportive regulatory frameworks for mobile health apps are further accelerating market expansion.
Get a Free Sample PDF Of This Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/download-sample/digital-biomarkers-market
Key Industry Developments
United States:
-February 2026: Biobeat announced the FDA clearance for its next-generation wearable biosensor platform, incorporating advanced digital biomarkers for real-time cardiovascular and respiratory monitoring, enhancing remote patient management with AI-driven predictive analytics. The technology integrates multi-sensor data fusion to detect early disease progression, supporting decentralized clinical trials and personalized treatment protocols. This launch addresses the growing demand for non-invasive, continuous health tracking in aging populations.
-January 2026: AliveCor unveiled an upgraded KardiaMobile 6L device with embedded digital biomarkers for atrial fibrillation detection and sleep disorder assessment, leveraging machine learning algorithms for improved accuracy. The product launch emphasizes integration with electronic health records for seamless clinician access, marking a key advancement in ambulatory cardiac diagnostics. It builds on prior FDA approvals to expand into neurological biomarker applications.
-December 2025: Verily (Alphabet Inc.) released R&D findings on its wearable platform's new gait analysis digital biomarker module, designed for neurodegenerative disease monitoring through precise motion and balance metrics. The development incorporates edge computing for real-time processing, positioning it for integration into large-scale Parkinson's and Alzheimer's studies. This initiative aligns with U.S. trends in precision neurology via sensor-based endpoints.
Japan:
-January 2026: Omron Healthcare launched a next-generation blood pressure monitor featuring embedded digital biomarkers for sleep apnea risk assessment, utilizing multi-sensor fusion and machine learning for proactive hypertension management. Targeted at Japan's elderly demographic, the device supports home-based interventions aligned with national health policies. This product advances continuous vital sign monitoring with predictive health insights.
-December 2025: Sony Corporation introduced an R&D prototype of biosensor-integrated earbuds capturing gait and balance digital biomarkers for elderly fall prevention, powered by edge AI for instant feedback. The technology facilitates home rehabilitation by analyzing real-world mobility patterns, contributing to Japan's aging society initiatives. It represents a leap in wearable audio-health convergence.
-November 2025: Panasonic Corporation advanced its healthcare division with a new AI platform for respiratory digital biomarkers, derived from smart spirometry devices for chronic disease tracking. The R&D breakthrough enables remote lung function monitoring with anomaly detection, tailored for Japan's telemedicine expansion. This supports early intervention in respiratory conditions prevalent among seniors.
Key Merges and Acquisitions:
-Medidata (Dassault Systèmes) – MC10 Digital Biomarker Business Acquisition (July 2025)
Medidata, a Dassault‑Systèmes‑owned clinical‑data and trial‑platform provider, acquired MC10’s digital biomarker business to integrate clinical‑grade biometric data and analytics from MC10’s BioStamp sensors into its Patient Cloud platform, strengthening its remote‑trial and digital‑biomarker offering for life‑sciences clients.
-Apple – Strategic Investment in Xandros (Digital‑Biomarker‑Focused Health Startup, March 2025)
Apple secured a USD 300 million strategic investment in Xandros, a digital‑health company specializing in digital biomarkers, to expand Apple’s health‑offering portfolio and deepen integration of validated digital biomarkers into its devices and consumer‑health services.
-AstraZeneca – Acquisition of Modella AI (AI‑Driven Oncology & Biomarker Discovery, details announced in early 2026 but transaction finalized in 2025)
AstraZeneca announced the acquisition of Modella AI, an AI‑driven oncology‑R&D platform company, following a multi‑year agreement in July 2025; the deal integrates Modella’s generative and agentic AI engine into AstraZeneca’s oncology R&D to accelerate clinical‑development timelines and enhance digital‑biomarker‑driven patient stratification and trial design.
Key Players:
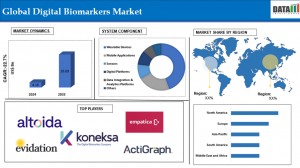
Koneksa Health | Altoida Inc | Empatica Inc | Evidation Health, Inc. | ActiGraph LLC | Quartet Health | IXICO plc | Vivo Sense | Aural Analytic | Neurotrack Technologies, Inc
Key Highlights: Top 5 Players in Digital Biomarkers Market 2026
-Koneksa Health: Advanced the K platform with AI-driven respiratory digital biomarkers for asthma and COPD clinical trials, enabling real-time remote monitoring and personalized treatment efficacy assessment through continuous physiological data analysis.
-Altoida Inc: Launched the Altoida Brain Health Platform featuring predictive cognitive decline biomarkers derived from AR-based neurocognitive assessments, supporting early Alzheimer's detection and intervention via machine learning-validated performance metrics.
-Empatica Inc: Expanded the Empatica Health Monitoring Platform with FDA-cleared cardiac digital biomarkers including continuous pulse rate and respiratory rate tracking on EmbracePlus wearables, facilitating longitudinal cardiovascular research and patient stratification in clinical studies.
-Evidation Health, Inc.: Introduced the Evidence Platform's Achievability engine with behavioral digital biomarkers for real-world evidence generation, integrating wearable and app data to quantify health outcomes and engagement in decentralized trials.
-ActiGraph LLC: Upgraded the CentrePoint platform with enhanced activity and sleep digital biomarkers featuring algorithmic refinements for wearable accelerometry, delivering precise physical function endpoints for pharmaceutical trials in neurology and metabolic disorders.
Buy Now & Unlock 360° Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=digital-biomarkers-market
Market Drivers and Key Trends:
-Remote Patient Monitoring: Digital biomarkers enable real-time health data collection via wearables, reducing hospital visits and supporting chronic disease management amid rising telemedicine adoption.
-AI and Machine Learning Integration: Advanced algorithms analyze biomarker data for predictive diagnostics, accelerating drug discovery and personalized medicine in pharma and healthcare sectors.
-Aging Population Pressures: Growing demand for early detection of neurodegenerative diseases like Alzheimer's through smartphone-based cognitive and mobility biomarkers.
-Regulatory Momentum: FDA approvals for digital biomarkers (e.g., in neurology and cardiology) validate their clinical utility, boosting investor confidence and market entry.
-Data Privacy Advances: Blockchain and federated learning trends address GDPR/HIPAA concerns, enabling secure sharing of vast multimodal datasets from apps and sensors.
Regional Insights:
-North America: 58.45% (Largest share, driven by advanced healthcare infrastructure, high adoption of wearables, and significant R&D investments in the U.S. and Canada).
-Asia Pacific: 25% (Fastest growing, fueled by rapid digital health adoption, government initiatives, and rising chronic disease prevalence in China, India, and Japan).
-Europe: 22% (Supported by strong regulatory frameworks like EMA guidelines, EU-funded R&D, and integration in chronic disease management).
Market Opportunities & Challenges: Digital Biomarkers Market 2026
Digital Biomarkers Market sees accelerating adoption through wearables and AI-driven remote monitoring, fueled by regulatory nods for digital endpoints in clinical trials. Growth hinges on real-time health data from sensors enhancing chronic disease management and decentralized trials.
-Opportunities
Wearables generate real-time biomarkers for efficacy tracking, boosted by sensor advancements enabling precise data fingerprinting.
Insurance firms leverage digital endpoints to streamline claims validation for chronic treatments, spurring payer adoption.
Integration into decentralized clinical trials cuts site visits, accelerates drug approvals via continuous endpoints.
Remote patient monitoring reduces hospital dependency, aligning with sustainable healthcare via fewer resources.
-Challenges
Cybersecurity risks expose sensitive biomarker data to theft and breaches, eroding trust.
Sample collection inconsistencies hinder reliable processing and validation.
Privacy regulations complicate data sharing in global trials.
-Strategic Verdict
AI-wearable hybrids and decentralized trial endpoints power 2026 dominance for chronic care innovators.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/digital-biomarkers-market
Market Segmentation Analysis:
-By System Component: Wearable Devices Lead with Ubiquity
Wearable devices command 45% market share in 2024, prized for real-time vital tracking in daily life via smartwatches and fitness bands.
Mobile applications hold 25%, enabling app-based symptom logging and AI-driven insights on smartphones.
Sensors capture 15%, embedded in devices for precise physiological data like heart rate.
Digital platforms and data integration & analytics platforms share 10%, unifying data for advanced analytics; others take 5% with emerging tools.
-By Application: Neurological Disorders Dominate Amid Aging Trends
Neurological disorders seize 30% share, propelled by Alzheimer's and Parkinson's monitoring needs in rising elderly demographics.
Cardiovascular diseases follow at 25%, leveraging biomarkers for arrhythmia and heart failure detection.
Respiratory disorders claim 20%, metabolic 15%, gastrointestinal 5%, and others 5%, addressing chronic condition tracking.
-By End User: Pharmaceutical Companies Spearhead R&D
Pharmaceutical companies lead with 35% share, investing in biomarkers for drug trials and personalized therapies.
Healthcare providers hold 25%, using them for patient monitoring in clinics.
Payers take 20%, academic & research institutions 15%, and others 5%, focusing on cost savings and innovation.
Conclusion:
The Digital Biomarkers Market is set to revolutionize healthcare by enabling proactive, data-driven patient management and accelerating drug development through AI-powered insights. As integration with wearables and regulatory support grow, this sector promises transformative outcomes in personalized medicine and chronic disease care by 2033.
Related Reports:
1. Cancer Biomarker Market - is expected to witness lucrative growth by reaching up to USD 32.5 billion by 2031.
2. Renal Biomarkers Market - is expected to grow with a high CAGR during the forecast period (2024-2031).
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
email us here
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.